Background: Autologous adoptive immunotherapy using T cells engineered with Chimeric Antigen Receptors (CAR-T cells) targeting CD19 and BCMA have demonstrated impressive results in B-cell malignancies, achieving long-term responses. However, similar results have not been obtained in other hematological diseases such as acute myeloid leukemia (AML). The aim of our study was to develop an optimized CAR-T product for the treatment of patients with R/R AML, that could address some of the current limitations associated with the lack of fitness of T-cells from patients with AML.
Methodology: Peripheral blood samples were collected from patients diagnosed with AML and from young and aged matched healthy donors. Second generation CAR-T developed based on 2 different antibodies and modifications in the co-stimulatory and hinge domains were tested using reporter cell lines. Phenotypic and functional characterization of CAR-T generated from controls and AML T-cells were performed. CRISPR-Cas9 technology and the Sleeping Beauty transposon system were employed to generate CAR-T cells with HLA-I and TCR knockout (HLA-I KO/TCR KO).
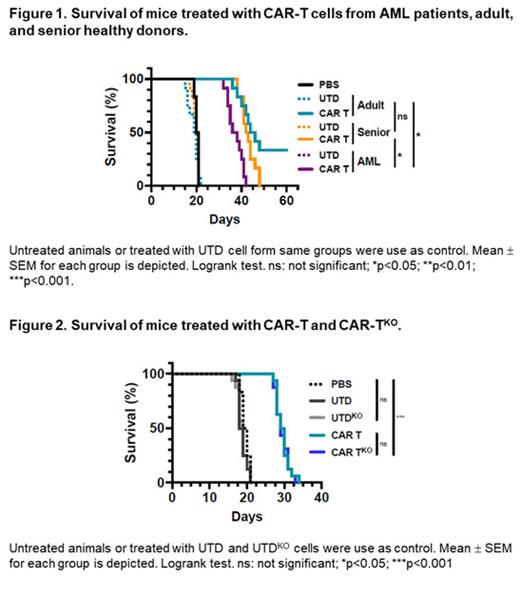
Results: We initially compared the phenotype of T cells from AML patients and healthy young and elderly controls. This analysis showed that T cells from AML patients displayed a predominantly effector phenotype, with increased expression of activation (CD69 and HLA-DR) and exhaustion markers (PD1 and LAG3), in contrast to the enriched memory phenotype observed in healthy donors. This differentiated and more exhausted phenotype was also observed, and corroborated by transcriptomic analyses, in CAR-T cells from AML patients engineered with an optimized CAR construct targeting CD33, resulting in a decreased in vivo antitumoral efficacy evaluated in xenograft AML models.
To overcome some of these limitations we combined CRISPR-based genome editing technologies with virus-free gene-transfer strategies using Sleeping Beauty transposons, to generate CAR-T cells depleted of HLA-I and TCR complexes for allogeneic approaches. Our optimized protocol allows one-step generation of edited CAR-T cells that show a similar phenotypic profile than non-edited CAR-T cells, with equivalent in vitro and in vivo antitumoral efficacy. Moreover, genomic analysis of edited CAR-T cells revealed a safe integration profile of the vector, with no preferences for specific genomic regions, with a highly specific editing of the HLA-I and TCR, without significant off-target sites. Finally, production of edited CAR-T cells at a larger scale allowed the generation and selection of enough HLA-IKO/TCRKO CAR-T cells that would be compatible with clinical applications.
Conclusions: In summary, our results demonstrate that CAR-T cells from AML patients, although functional, present phenotypic and functional features that could compromise their antitumoral efficacy, compared to CAR-T cells from healthy donors. The combination of CRISPR technologies with transposon-based delivery strategies allow the generation of HLA-IKO/TCRKO CAR-T cells, compatible with allogeneic approaches, that would represent a promising option for AML treatment.
Disclosures
Pierola:Novartis: Speakers Bureau; BMS: Consultancy, Speakers Bureau; Abbvie: Speakers Bureau; Jazz Pharma: Consultancy, Speakers Bureau; Syros: Consultancy, Speakers Bureau; Astellas: Consultancy; Astra Zeneca: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal